Preliminary Assessment of Common Vitamins
A Simple Elemental Chemical Test to find out the truth.
Introduction
In March 2023, I was made aware of the massive scam that is vitamins. Unearthing this scam resulted in me receiving a lot of kick back from people who we thought were of a similar mind but, as it turns out, there are a lot of influencers in the so-called “medical freedom movement” who promote vitamins as a sideline income. Some articles we have written on the vitamin scam in the past include:
In collaboration with Agent131711, we have decided to carry out some of our own experiments on vitamins, starting with a simple elemental chemical test. The aim being to verify our previous claims for ourselves and our readers.
This particular experiment began with me going to the local pharmacy, choosing and purchasing three different brands of Vitamin B products to submit for an elemental chemical test (testing was funded by Agent131711).
Elemental analysis is a process in analytical chemistry in which a sample of material such as water, minerals or bodily fluid is analyzed to ascertain its elemental composition. Elements being those that appear on the periodic table - pure substances that cannot be broken down by any physical or chemical means into simpler substances. An element is made up of the same kind of atoms.
Elemental analysis can be qualitative, determining which elements are present or quantitative, determining the levels of elements.
In this case we opted for a quantitative elemental analysis. What this means is that the tests would quantify the level of certain elements in the sample, and it wouldn't determine every element that is present.
To enable us to continue carrying out experiments like these please consider buying me a coffee or becoming a member of my “by me a coffee” channel. This will help us fund the experiments as well as motivate the authors writing these articles.
Sample Preparation
250 ml sample bottles were purchased, and the experiment was laid out before we began with sample preparation. Figure 1 and Figure 2 below shows the labelling and materials used for this experiment.
As can be seen from Figure 1 below, we submitted four samples for elemental analysis. Sample 1 was to function as our control and consisted of only the water in which the vitamins in the other samples would be dissolved. This allowed us to ascertain what quantity of the elements detected should be attributed to the water and which should be attributed to the vitamin samples.
Each of the samples had to be dissolved in water for the elemental analysis and could not be submitted as is. Three pills of each vitamin in the respective samples were dissolved in 250 ml of water in brand new plastic bottles.
The decision was made to dissolve three pills into each sample in order to ensure that there was enough vitamin sample to be detected in the test. We were not certain that dissolving the recommended daily intake of 1 pill in the water solution would produce results too small for detection and the analysis would be a waste of time and money. Particularly because the methods employed for dissolving the pill contents did not accurately simulate what would take place in a person's stomach (increased temperature and stomach acid would dissolve the pills better). The expected dose will be tested in the next experiments (dissolving 1 pill in water vs dissolving 1 pill in warm water with acid added to simulate stomach conditions).
Note: I attempted to dissolve Sample 3 and Sample 4 pills in the water (water was room temperature) but after a few minutes of shaking the bottles I realized that the pills were not dissolving. As such, I crushed the pills on a clean plate with a spoon and added this powder to the sample bottles filled with water (the same water that was tested as a control).
Sample 2 preparation.
The first specimen tested is High B Vitamin B Complex (High Potency) and an image of this product can be seen below.
These vitamins are in capsule form and as such the three capsules were opened and the contents of each emptied into the control water. It was given a proper shake before it was submitted to the lab for analysis. Note that in addition to vitamin B1, 2, 6 and 12 this product also contains calcium pantothenate.
Sample 3 preparation.
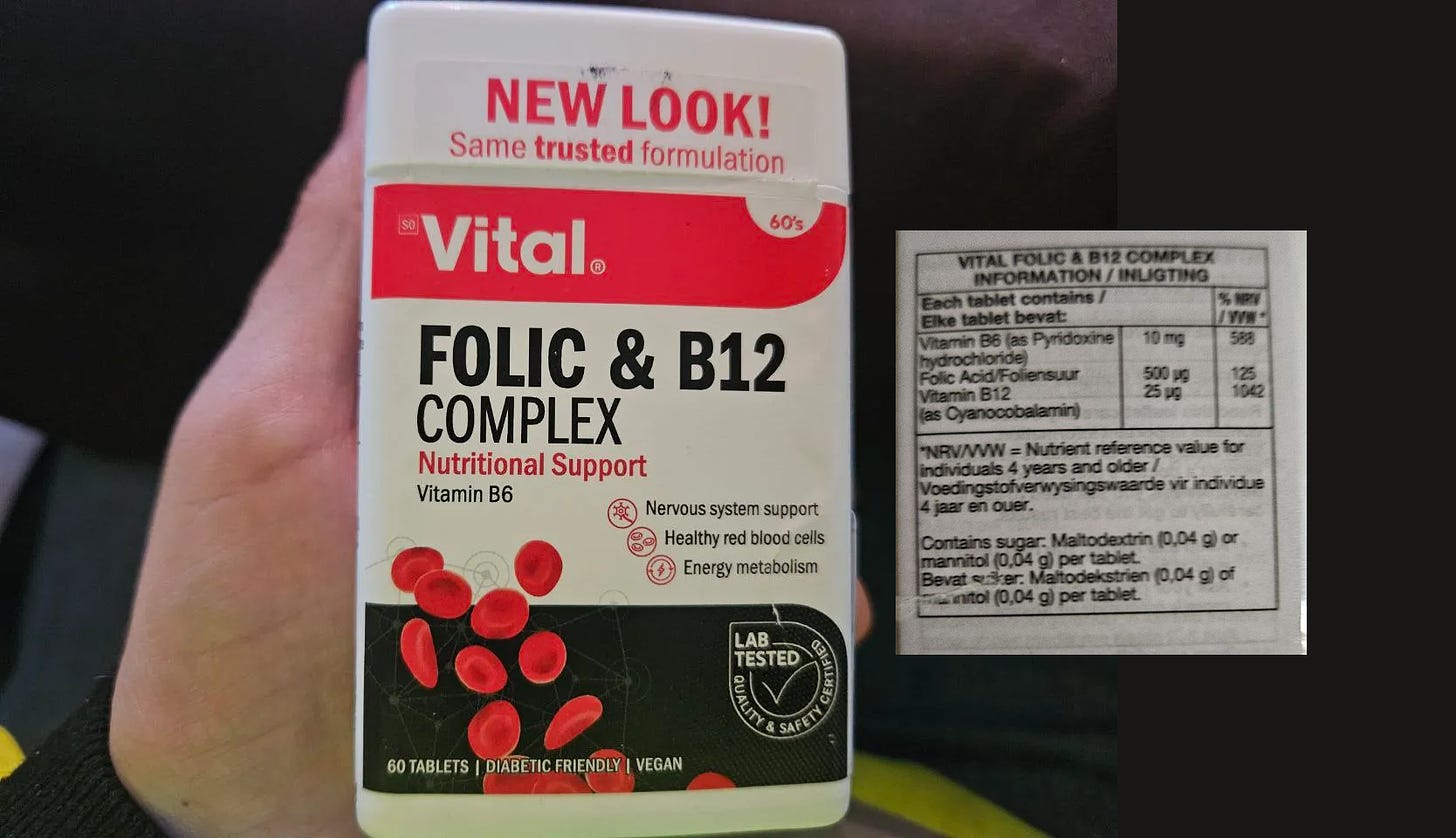
The second specimen tested is Folic & B12 Complex (Nutritional Support) from Vital and an image of this product can be seen below.
The product consists of pills that did not easily dissolve in the control water. To get around this issue, I opted to crush the three pills with a spoon against a clean plate and add the crushed pill powder to the control water.
Sample 4 preparation.
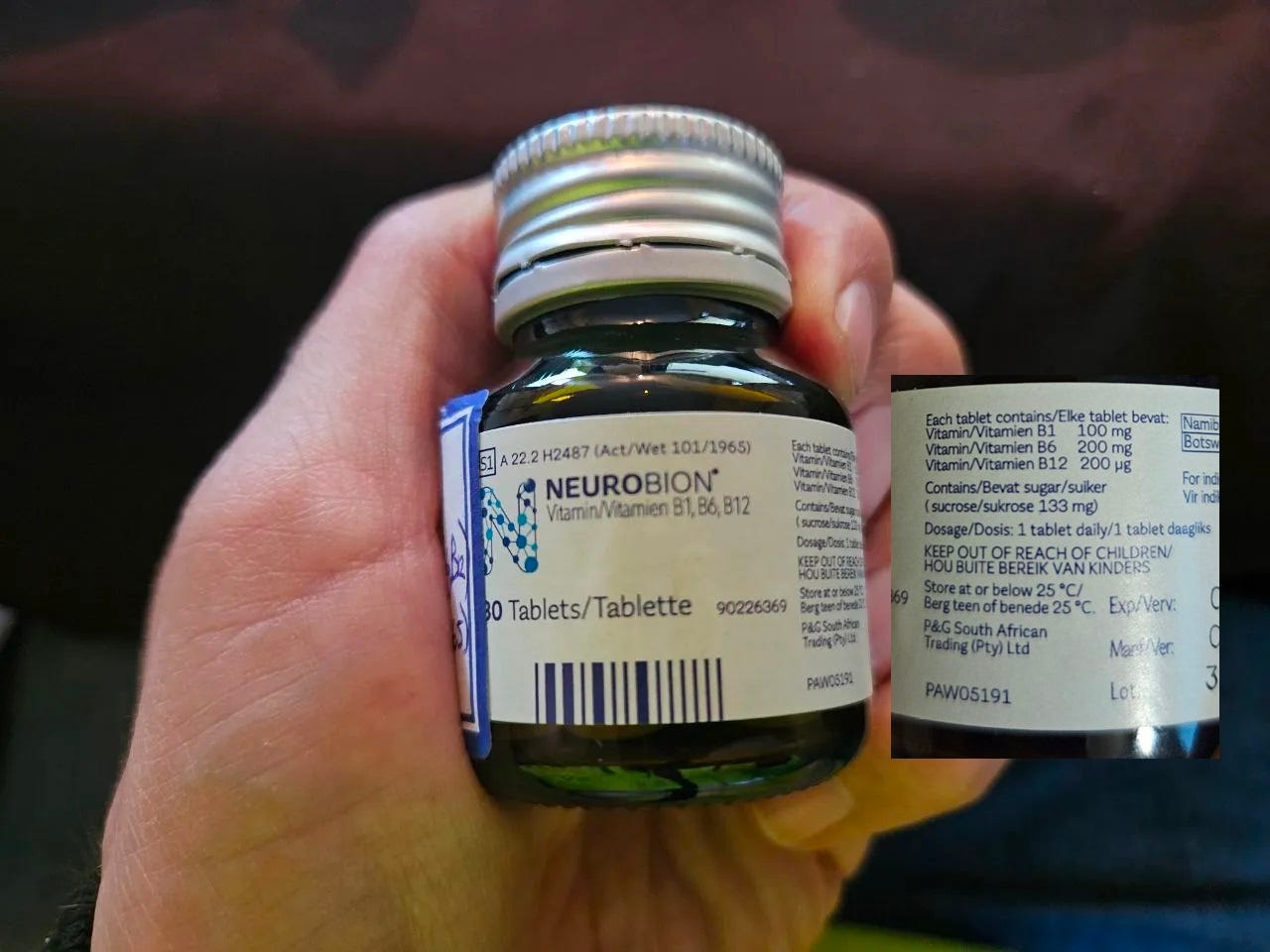
The third specimen tested is Neorobion Vitamin B1, B6, B12 and an image of this product can be seen below.
Similar to sample 3, this product also consisted of pills that did not easily dissolve in the control water. So as with sample 3, these pills were also crushed with a clean spoon against a clean plate and the resultant powder added to the control water.
The samples were then boxed, and hand delivered to the lab in a box as shown in the below image.
This particular company typically performs elemental analysis on drinking water, which is then measured against the recommended acceptable levels based on recognized guidelines. As such, this analysis is not necessarily determining toxicity, but it is a good screening method to establish what is actually in the samples. Once we know which elements are elevated, we can further look at the potential toxicity due to the chemicals involved.
Expected Substances
The ingredients of each of the samples are listed below:
Sample 1:
Sample 1 is the control. This is the water only sample which was put in the same plastic bottle as all the other samples. If there was anything in the water to start off with or the plastic from the bottle leached into the water, it would be picked up in this control sample. Further, anything in the control water would appear equally across all the samples and as a result easily attributable to the water used not the sample.
Sample 2 - Each capsule contains:
Vitamin B1 (Thaimin HCL).
Vitamin B2 (Riboflavin).
Vitamin B6 (Pyridoxine HCL).
Vitamin B12 (Cyanocobalamine).
Calcium Pantothenate
Sample 3 - Each capsule contains:
Vitamin B6 (Pyridoxine HCL).
Folic Acid (vit B 9).
Vitamin B12 (Cyanocobalamine).
Sample 4 - Each capsule contains:
Vitamin B1 (*NS).
Vitamin B6 (*NS).
Vitamin B12 (*NS).
*NS - Not Specified
We know from Agent131711's vitamin articles that the chemicals (and their chemical formulas) which have been deemed vitamin B are as follows. As such, we can expect the elements which make up these chemicals to show up in the resutls:
Vit B 1 =Thiame (C12H17N4OS+)
Vit B 2 = Ribonflavin (C17H20N4O6)
Vit B 6 = Pyridoxine (C8H11NO3)
Vit B 9 = folic acid (C19H19N7O6)
Vit B 12 = Cyanocobalamine (C63H88CoN14O14P)
For a more detailed post on Vitamin B specifically please refer to Agent’s post titled: "Vitamin B" Contaminated a City: The Time a Vitamin Manufacturer Destroyed a Town, Caused Cancer, Birth Defects, Illness.
Based on the above chemical formulas for the different ingredients, we would expect to find various quantities of the following elements in the samples:
C - Carbon
H - Hydrogen
N - Nitorgen
O - Oxygen
S - Sulfur
Co - Cobalt
P - Phosphorus
Ca - Calcium (from the calcium Pantothenate)
Note - Even though all the above elements should be present in the samples only those chemicals in bold were tested for and quantified in the analysis. In other words, the analysis did not quantify how much carbon, hydrogen, Nitrogen etc featured in the samples.
Therefore, and in theory, the only elements which should appear in the results in addition to those in the control water are cobalt, calcium and phosphorus.
It's important to realize that the results of this analysis only provides a list and level of certain elements present in the samples - in particular those elements that should not be found above a certain level in our drinking water.
As such, the results should not be taken to demonstrate whether there are chemicals in the samples. Elements are the building blocks of chemical molecules - the samples were not analyzed for the chemical molecules themselves. However, by doing an elemental analysis first we are put in a better position to know what chemicals to test the samples for in the future.
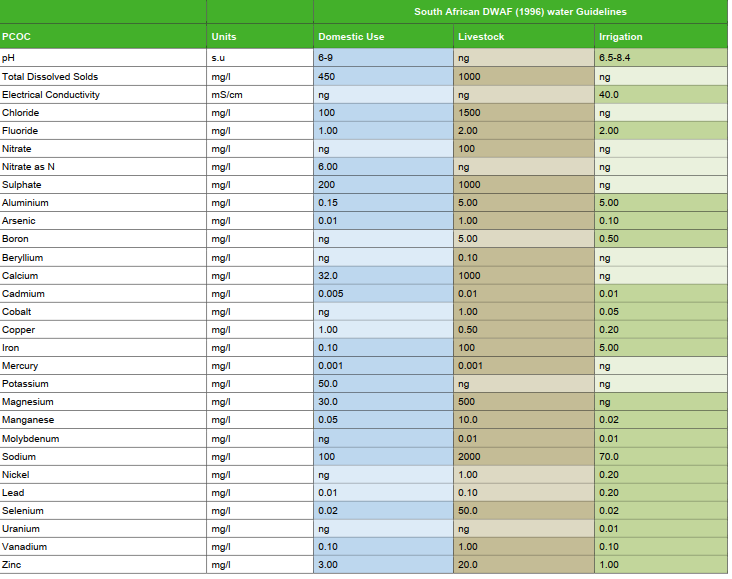
Acceptable limits
There are various different guidelines that can be used to verify limits for different elements, but the guidelines used for this article are the South African regulations for water for domestic use. Figure 6 below shows the different limits for different elements.
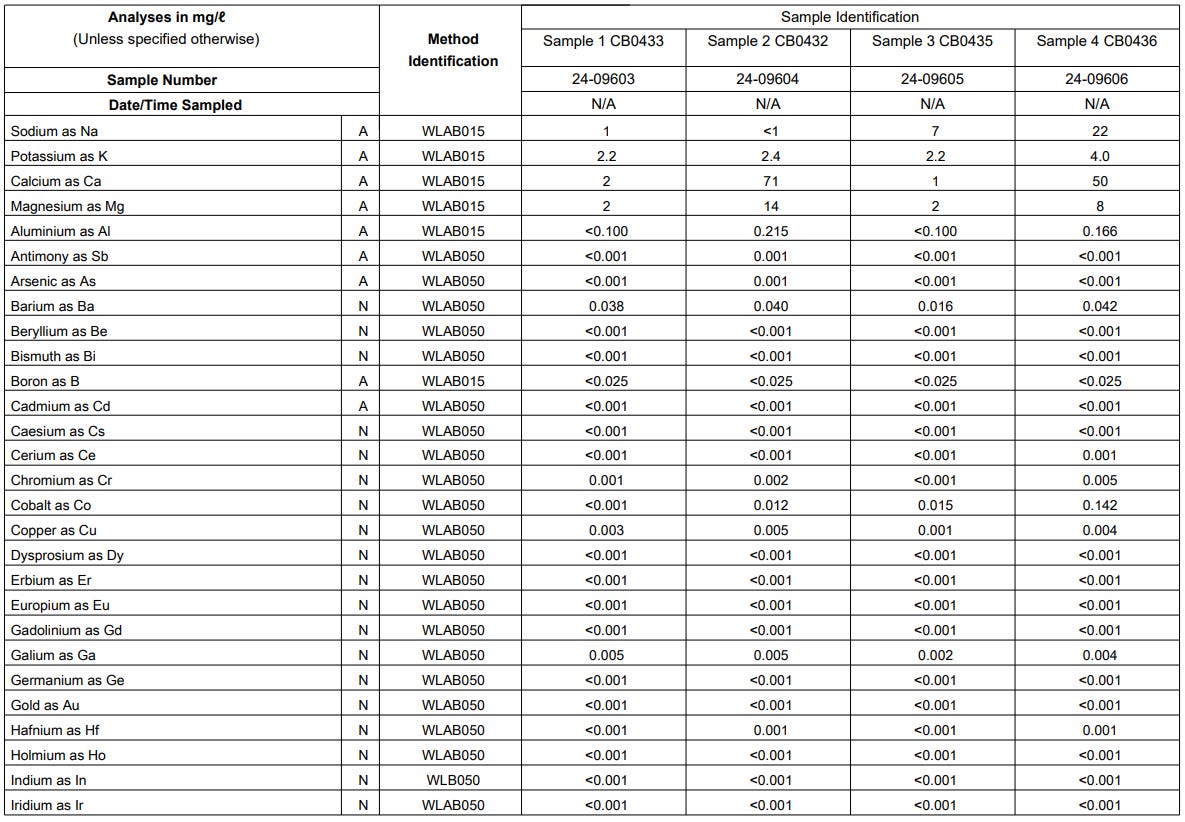
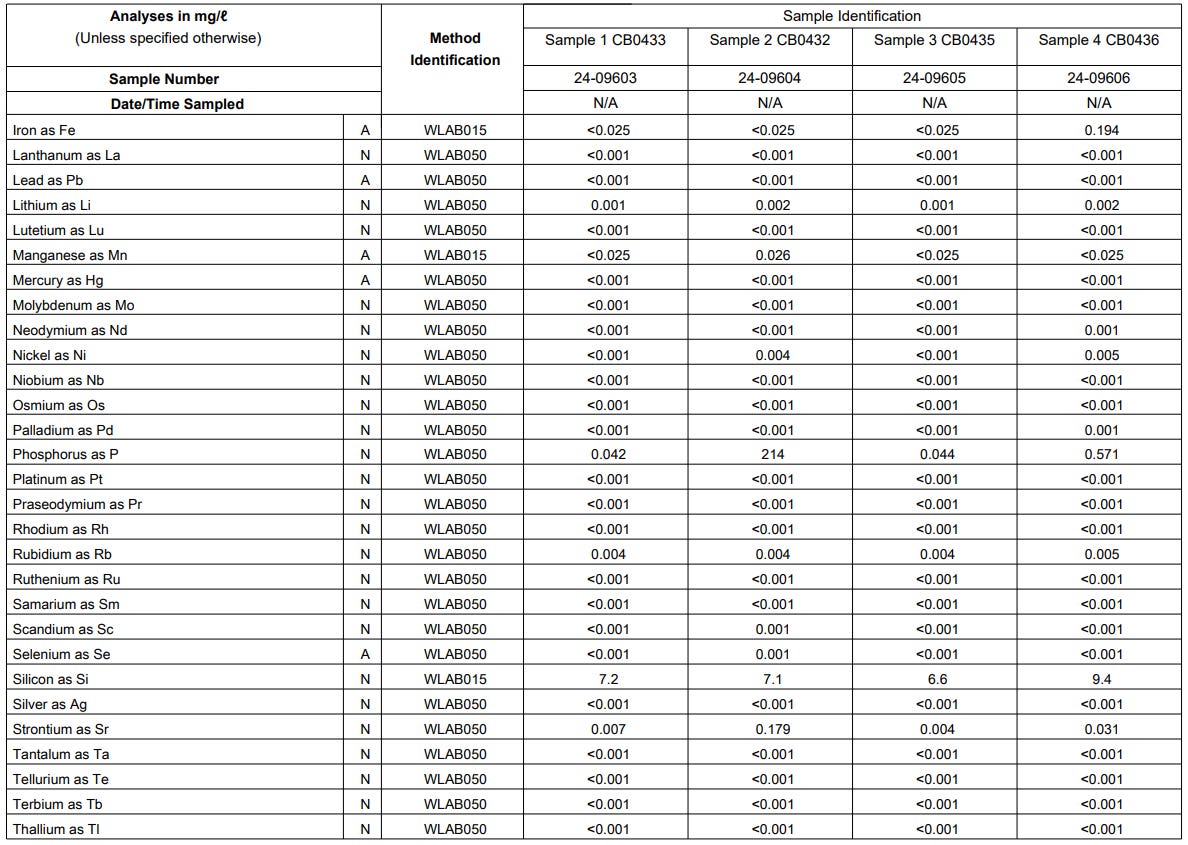
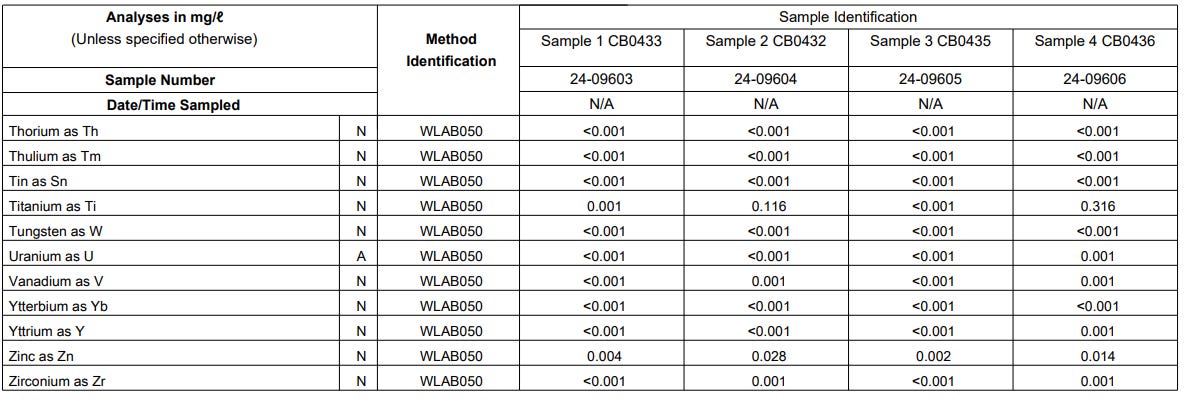
Results
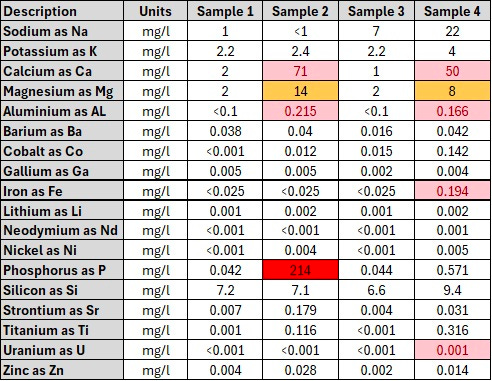
The results obtained from the elemental analysis conducted in the different samples are shown in Figure 7, Figure 8 and Figure 9 below.
Results Discussion
The following elements were detected and although we do not have a limit on all of these elements yet, we intend to do further research to see how toxic these elements are (if at all):
The cells highlighted in light red exceeded the South African Regulatory guideline limits on drinking water.
Cells highlighted in orange are close to these limits.
The cell highlighted in red was a reading that was picked up on Phosphorus (P) which has no limit according to the South African Regulatory guideline limits on drinking water.
Sodium (Na) is detected in the control sample, but Sample 3 and Sample 4 do show elevated level of Sodium. The recommended limit for Sodium is discussed further down in this article.
Potassium (K) is elevated in the control and is fairly consistent for all samples which means that the reading for all samples is most likely due to the Potassium in the control water. Sample 4 however does show an elevated reading of Potassium which could be investigated further.
Calcium (Ca) is elevated in the control which means it will be present in all the samples. The level of Calcium in sample 2 and Sample 4 however, is elevated even beyond the recommended limits (refer to cells highlighted light red in Table 1). Ingestion of this chemical is discussed further down in this article.
Magnesium (Mg) is elevated in the control which means it will be present in all samples. The level of Mg in Sample 2 and Sample 4 however, is elevated and is close to the recommended limits (refer to cells highlighted light red in Table 1). Ingestion of this chemical is discussed further down in this article.
Aluminum (Al) is present in Sample 2 and Sample 4 even higher than the recommended levels (ingestion of this chemical is discussed further down in this article).
Barium (Ba) is present in the control water and shows the same level for all samples which means that it was just the Barium levels in the control water that was detected.
Cobalt (Co) is expected to be elevated for samples containing B12. Sample 2 and Sample 3 do show that Cobalt is present and Sample 4 shows even higher levels of Cobalt (ingestion of this chemical is discussed further down in this article).
Gallium (Ga) is present in the control water and shows the same level for all samples which means that it was just the Gallium levels in the control water that was detected.
Iron (Fe) is elevated for Sample 4 even beyond the recommended dose for drinking water. No Iron was picked up in any of the other samples.
Lithium (Li) is present in the control water and shows the same level for all samples which means that it was just the Lithium levels in the control water that was detected.
Neodymium (Nd) is detected in Sample 4 although it is in very small quantity. Perhaps a retest considering the recommendations will give a better indication in the next round of testing.
Nickel (Ni) is detected in Sample 2 and Sample 4 although it is in very small quantity. Perhaps a retest considering the recommendations will give a better indication in the next round of testing.
Phosphorus (P) is expected to be in the results. The presence of this chemical in the samples should be further investigated especially for Sample 2 which shows a high concentration of this element.
Silicon (Si) is present in the control water and shows the same level for all samples which means that it was just the Silicon levels in the control water that was detected.
Strontium (Sr) is elevated in Sample 2 although it is only in a small quantity. Perhaps a retest considering the recommendations will give a better indication in the next round of testing.
Titanium (Ti) is elevated in Sample 2 and Sample 4 although it is only in a small quantity. Perhaps a retest considering the recommendations will give a better indication in the next round of testing.
Uranium (U) is detected in Sample 4 but only in a very small quantity. Perhaps a retest considering the recommendations will give a better indication in the next round of testing.
Zinc (Zn) is detected in Sample 2 and Sample 4 but only in a very small quantity. Perhaps a retest considering the recommendations will give a better indication in the next round of testing.
Unexpected Elements
Another way to look at the results can be set out as follows:
Given the listed ingredients of sample 2, we expected to find traces of cobalt, phosphorus and calcium. What we did not expect to find were traces of magnesium, aluminum, nickel, strontium, titanium and zinc.
Given the listed ingredients of sample 3, we expected to only find traces of cobalt and phosphorus both of which didn't really feature in the results. Then aside from slightly elevated levels of Barium, nothing unexpected was found.
Given the listed ingredients of sample 4, we expected to only find traces of cobalt and phosphorus. What we did not expect to find were traces of calcium, magnesium, aluminum, barium, iron, nickel, silicon, uranium, strontium, titanium and zinc.
Some of the elements detected in this experiment are discussed further below in order to ascertain whether these elements pose a risk to health or not.
Note - Many of these amounts may seem insignificant but it is important to remember that it is recommended that you take these pills on a daily basis.
Sodium
Sample 4 show an elevated level of Sodium and according to Oregon Department of Human Services, Health Effects Information the following was concluded on Sodium:
There is no standard for sodium in drinking water at the federal level, but USEPA recommends that drinking water sodium be held to 20 ppm or less because sodium is so common in other beverages and food.
Note - The conversion from ppm to mg/l is 1 to 1. 20 ppm is therefore 20 mg/l. Sample 4 exceeds this limit and sample 3 comes close to the limit.
Potassium
WHO has a limit on an acceptable level of potassium in drinking water as follows (refer to this link):
According to WHO standards the permissible limit of potassium is 12 mg/1.
Calcium
Sample 2 and Sample 3 show calcium levels exceeding the limit of 30 mg/l. Sample 2 actually exceeds this limit by double the recommended limit. We can consider the effects of dangerous levels of calcium by reviewing this article (Hypercalcemia). Some highlights from the article as follows:
Kidneys. Excess calcium makes the kidneys work harder to filter it. This can cause serious thirst and frequent urination.
Digestive system. Hypercalcemia can cause stomach upset or pain, vomiting, and constipation.
Bones and muscles. Most often, the extra calcium in the blood is leached from the bones. This weakens the bones. It can cause bone pain and muscle weakness.
Brain. Hypercalcemia can affect how the brain works. That can lead to trouble focusing, confusion, drowsiness and fatigue. It also can cause depression.
Heart. Rarely, serious hypercalcemia can affect the heart. It can cause feelings of a fast beating, fluttering or pounding heart. It also can cause the heart to beat out of rhythm. It's linked with other heart-related conditions as well.
Although the levels of calcium in the vitamins (as shown in the results of this article) are not an indication of levels of calcium in the blood, one can get a good idea at what level calcium becomes dangerous. The paper by M. Nazia, 2024. Hypercalcemia outlines the different levels in the blood as follows:
Mild hypercalcemia: 10.5 to 11.9 mg/dL
Moderate hypercalcemia: 12.0 to 13.9 mg/dL
Hypercalcemic crisis: 14.0 to 16.0 mg/dL
One should also consider the fact that when these guidelines set limits, they only consider the effect of a single substance. They never consider the combined effects that all these substances have on the body when taken together. I therefore want to emphasize that these limits could actually be much higher than what they should be.
Aluminum
The toxicity of Aluminum is well known, and it is commonly used in vaccinations. Although the damage done by this substance through ingestion is not nearly as severe as the damage it causes through injection its effects are still a concern. Some characteristics of Aluminum (Al) is described by L. Tomljenovic, 2010. Aluminum and Alzheimer's disease: after a century of controversy, is there a plausible link? as follows:
Very small amounts of Al are needed to produce neurotoxicity and this criterion is satisfied through dietary Al intake
Al sequesters different transport mechanisms to actively traverse brain barriers
Incremental acquisition of small amounts of Al over a lifetime favors its selective accumulation in brain tissues
Since 1911, experimental evidence has repeatedly demonstrated that chronic Al intoxication reproduces neuropathological hallmarks of AD.
Further to the above I. O. Igbokwe, 2019. Aluminium toxicosis: a review of toxic actions and effects explains the following about Aluminum:
Toxic actions of Al induce oxidative stress, immunologic alterations, genotoxicity, pro-inflammatory effect, peptide denaturation or transformation, enzymatic dysfunction, metabolic derangement, amyloidogenesis, membrane perturbation, iron dyshomeostasis, apoptosis, necrosis and dysplasia. The pathological conditions associated with Al toxicosis are desquamative interstitial pneumonia, pulmonary alveolar proteinosis, granulomas, granulomatosis and fibrosis, toxic myocarditis, thrombosis and ischemic stroke, granulomatous enteritis, Crohn’s disease, inflammatory bowel diseases, anemia, Alzheimer’s disease, dementia, sclerosis, autism, macrophagic myofasciitis, osteomalacia, oligospermia and infertility, hepatorenal disease, breast cancer and cyst, pancreatitis, pancreatic necrosis and diabetes mellitus.
Also see the work by Chris Exley here.
Iron
According to Guidelines for drinking-water quality, 2nd ed. Vol. 2. Health criteria and other supporting information. World Health Organization, Geneva, 1996 the following can be concluded:
As a precaution against storage of excessive iron in the body, JECFA established a provisional maximum tolerable daily intake (PMTDI) in 1983 of 0.8 mg/kg of body weight (14), which applies to iron from all sources except for iron oxides used as colouring agents, and iron supplements taken during pregnancy and lactation or for specific clinical requirements. Allocation of 10% of this PMTDI to drinking-water gives a value of about 2 mg/litre, which does not present a hazard to health. The taste and appearance of drinking water will usually be affected below this level, although iron concentrations of 1–3 mg/litre can be acceptable for people drinking anaerobic well-water. No health-based guideline value for iron is proposed.
According to the above memo a “recommended” acceptable level of iron is 56 mg/day. The South African regulatory guideline seems to be more a guide for taste and color as opposed to health reasons.
It is however important that we consider the effects of Iron toxicity. According to Symptoms & Causes of Hemochromatosis the buildup of harmful levels of iron, hemochromatosis, can cause symptoms including:
Feeling tired or weak
Pain in the joints, particularly in the knees and hands
Loss of interest in sex or erectile dysfunction
Darkening of skin color, which may appear gray, metallic, or bronze
With more severe iron overload, people may develop signs and symptoms of complications, such as cirrhosis, diabetes, or heart failure NIH external link.
Not everyone with hemochromatosis has symptoms, and hemochromatosis may not cause symptoms for many years. Symptoms typically begin after age 40, and, on average, women develop symptoms about 10 years later than men do.
Uranium
A very low level of Uranium was picked up in Sample 4 and it would probably be best if a re-evaluation of this substance be undertaken to confirm whether this reading is correct or not. Toxicological Profile for Uranium had the following about Uranium:
The toxicity of uranium varies according to its chemical form and route of exposure. On the basis of the toxicity of different uranium compounds in animals, it was concluded that the relatively more water-soluble compounds (uranyl nitrate, uranium hexafluoride, uranyl fluoride, uranium tetrachloride) were the most potent systemic toxicants.
Magnesium
Although the following relates to levels of magnesium in the blood rather than levels of magnesium ingested (which is the results of our experiment) according to what is hypermagnesemia the following guideline can be considered:
Hypermagnesemia is diagnosed using a blood test. The level of magnesium found in the blood indicates the severity of the condition.
A normal level of magnesium is between 1.7 and 2.3 mg/dL. Anything above this and up to around 7 mg/dL can cause mild symptoms, including flushing, nausea, and headache.
Magnesium levels between 7 and 12 mg/dL can impact the heart and lungs, and levels in the upper end of this range may cause extreme fatigue and low blood pressure.
Levels above 12 mg/dL can lead to muscle paralysis and hyperventilation. When levels are above 15.6 mg/dL, the condition may result in a coma.
Cobalt
According to V. Venkatraman, 2020. Cobalt-Induced Toxicity and Spasticity Secondary to Hip Arthroplasty: Case Report and Review of the Literature the following can be concluded on Cobalt toxicity:
Normal daily dietary intake ranges from 5 to 50 μg, with normal plasma concentrations of <0.2 μg/L. Excessive cobalt can produce systemic toxicity affecting multiple organ systems. Adverse effects related to cobalt toxicity can occur at levels of 7-10 μg/L or more.
Phosphorus
There are various forms of phosphorus to considered when looking at its toxicity. Princeton University Environmental Health and Safety concluded the following:
Amorphorous phosphorus (red phosphorus) is a reddish-violet powder that is not considered toxic in its pure form. It is stable under ordinary conditions, however is flammable and can be initiated by excesive shock or friction.
Yellow phosphorus (white phosphorus) is a significantly more hazardous form of the element and may be present as a contaminant in red phosphorus. This allotrope of phosphorus is extremely toxic, and the estimated human lethal dose is 50 - 100 mg. This form must be stored under water as it burns rapidly and is spontaneously combustible upon exposure to air.
When working with amorphous phosphorus, it is prudent to consider the potential for yellow phosphorus contamination and related precautions required to handle the material safely.
One of the main reasons for elevated phosphorus levels in water is due to fertilization of crops and although it can cause problems such as excessive growth of algae and aquatic plants this substance is generally not considered a pollutant. More information on phosphorus provided by Phosphorus and Water Quality are as follows:
Phosphorus is the plant nutrient most often responsible for impairment of surface water. It is essential for healthy, productive crops and most agronomic systems call for application of commercial fertilizer or animal manure to correct soil phosphorus deficiencies. For agronomic, economic and environmental reasons, application of phosphorus is not recommended, however, where soil tests show phosphorus reserves are adequate for the intended crop.
Phosphorus is typically not considered a groundwater pollutant. Further, most soils have the capacity to retain phosphorus in the upper layers, protecting the groundwater.
Experiment Improvement
Going forward we would like to send a different group of vitamin products for elemental analysis, for example a few prenatal vitamins products or simply a group of vitamin D products. In addition, we would like to improve on the results set out above.
One obvious improvement that can be included in the experiment is to find a method to better dissolve the specimens in the control water. It should be noted that the results obtained in this first round is most likely understated due to the difficulty of dissolving the pills/powder into the control water. Considering that the stomach is not only warmer than room temperature but also acidic, your body will absorb much more of the contents of these products than what is shown in the results. For the next round I will consider warming the control water or adding a slight acidic solution (or both) to assist with dissolving the product.
This method of elemental chemical testing was deemed to be the easiest and cheapest way to test the different samples. However, there are a lot of substances that can be missed by using this method given that most chemicals consist of carbon (C), hydrogen (H), Sulphur (S), chlorine (CL), Nitrogen (N) and oxygen (O) etc - in other words elements which are not tested for and quantified by elemental analysis. Some examples being Coal tar (C137H97O9NS), ammonia (NH3), acetone ((CH3)2CO), chloroform (CHCl3), GMO soy, GMO sugar, GMO mold derivatives, pesticides and typical lab made chemicals can be present in the samples and be missed in the results.
We would ideally like to send the samples for a chemical analysis - likely Chromatography and mass spectrometry. A list of additional chemicals of concern will be submitted as part of the testing requirements for the next round. Please add the chemicals you’d like to see tested for in the next round in the comments and we will consider them.
The level of elements being detected will also be considered for different samples (dissolving 1 pill in water vs dissolving 1 pill in warm water with acid added to simulate stomach conditions).
If someone is aware of a better test we can undertake that does not cost an arm and leg please let us know in the comments.
Conclusion
2 of the 3 products assessed in this experiment were shown to contain unsafe levels of Calcium, Magnesium, Iron and possibly Uranium according to the South African regulations for water for domestic use. Sample 4 also exceeds the accepted level of Sodium according to Oregon Department of Human Services, Health Effects Information.
In addition, the results show that sample 2 and 4 have the ingredients they are supposed (Co and P) to but also have extras that they shouldn't or which are not listed - we will then look into these extras. Sample 3 on the other hand doesn't show traces of what it should have (Co and P) but nor does it show anything else really - indicating sample 3 is probably just a sugar pill and not worth testing again.
Even though the levels seem low it should be noted that experimental conditions more representative of that experienced in the body could significantly impact the results which makes these products so much more unsafe for consumption (this might improve in further studies).
In addition, remember these vitamins are also recommended to be ingested daily for prolonged periods of time. This means that if your body is not able to get rid of these elements quickly it will start to build up within your body.
Although we are fully aware that synthetic vitamins are useless, this experiment was undertaken to further shown the potential harm these products pose to health.
Please be aware that we are not doctors or scientists but merely investigative journalists. We are trying to make sense of the data we get and may not be aware of certain nuances. If you think we have missed things or misinterpreted certain data please let us know in the comments.
Finally, if you take these products or products similar to these and are happy with how you feel, that's great. We are not trying to tell you what to do, we are merely presenting information that we believe important. What you do with this information is not really our business.
If you’d do like to support our work, consider buying me a coffee or becoming a member on my “by me a coffee” account.
Also see Jamie’s (@controlstudies) control experiment here.
Other great reads:
- - The Flawed Non-Specificity of Polio Diagnostics
- - Measles! What Is It Good For?
- - It matters that viruses don't exist
- - Dismantling Vitamin D-3
- - Proton Goes to the Virus Circus🎪
- - The Final Countdown (?)
















"Very small amounts of Al are needed to produce neurotoxicity and this criterion is satisfied through dietary Al intake"
-This is great DIY, amazing really.
-I wonder, can you get a Corona shot vial and test the elements in that? You need to get a nurse who works in a place they give vaxes to take a used vial out of the waste can with pinchers, there is always some fluid left in used vials.
Excellent as always, and thank you for having me help out a bit! Vitamins are a fantastic example of big pharma false dichotomy.